Key to Common Western Oregon Species of Rhizopogon
by Matt Trappe, revised 01-Oct-2011
Rhizopogons are among the most commonly encountered hypogeous fungi in western Oregon. They are found in a variety of habitats, from coastal dunes to subalpine forests. Their peridium ranges in color from white to almost black, with shades of dirty white to beige predominating. Some are bright yellow or apricot-colored, and many bruise brownish or red upon exposure to air or handling. The gleba color is also quite variable; from white (especially in immature specimens) to tan to dark olive-green to brown or grey. The gleba of Rhizopogons is usually firmly spongy, with small chambers and a consistent texture (not marbled or veined). Rhizopogon spores are relatively small, generally ellipsoid to bacilliform, and smooth. It should be stated up front that it is impossible to confidently identify Rhizopogon species without a microscope.
The only comprehensive key to North American species of Rhizopogon is the 1966 monograph by Alexander H. Smith & Sanford M. Zeller (Memoirs of the New York Botanical Garden, Vol. 14, No. 2). Alexander Smith undertook the intimidating task of sorting out this huge and baffling genus, and without his efforts we would be in a very bad muddle indeed (AHS gave SMZ co-authorship 18 years posthumously in recognition of SMZ's pioneering work on the genus). Subsequent study has revealed that some of Smith's descriptions were different developmental stages of the same species. The Smith & Zeller key also relies extensively on fresh characteristics and colors at different developmental stages, pieces of information that are all too often lacking when confronted with a dried specimen. Their treatment of species not found on the west coast further obfuscates identification of local taxa.
This key contains only the more common species of Rhizopogon found in western Oregon - it is far from comprehensive. It attempts to use only those characters apparent in dried collections and assumes no fresh notes. The section names follow Smith & Zeller but are used loosely, with several species moved or keyed repetitively, and the entire taxonomic section Fulviglebae eliminated. The key is primarily based on Smith & Zeller's descriptions, personal communications from experienced taxonomists (Drs. J. Trappe, D. Luoma, and M. Castellano - thank you!), and examinations of dried Pacific Northwestern collections - not holotypes and not fresh material. Parenthetic notations following species names are Smith & Zeller page references. Always check the detailed descriptions in Smith & Zeller to confirm identifications. This key is designed to be easier to use than Smith & Zeller's monograph, but it surely isn't perfect. Please email the author with feedback and suggestions for improvement.
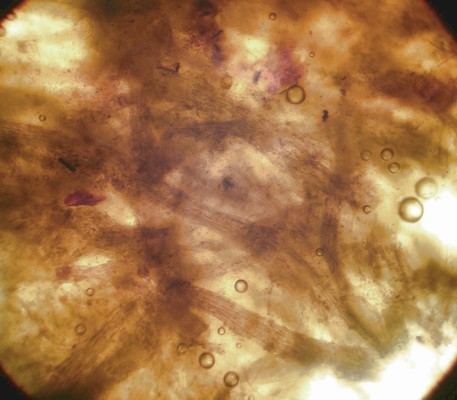
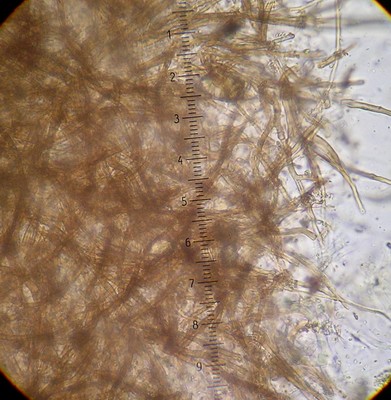
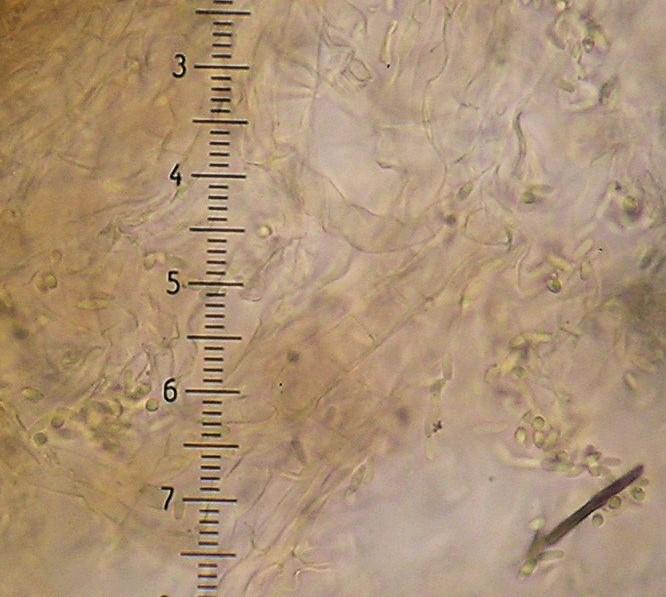
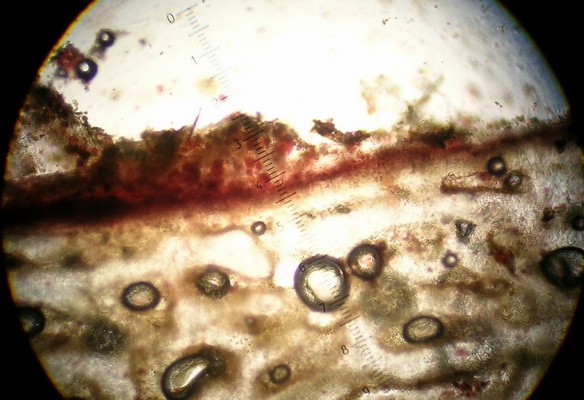
An important characteristic in Rhizopogon identification is the hyphal structure of the peridium. There are three basic types as viewed in 5% KOH mount: rhizomorphic cables that look like bundles of translucent rope throughout the peridium (Fig. 1), loosely woven brown-walled hyphae (Section Villosuli; Fig. 2), or tightly woven hyphae that are hyaline to orangish-brown (Fig. 3) or with red pigments. Sometimes this tightly woven peridium type has so much debris incorporated that the structure resembles chipboard (Fig. 4).
To observe peridial structure, take thin sections from the edge of the specimen in several places (Fig. 5; some section easier if first wetted with KOH, others section best when dry), place them on a slide with a drop of KOH, add cover slip, and bounce the eraser end of a pencil on the slip several times from about an inch up. This splays out the sections and facilitates observation of peridial hyphae (Fig. 6).
Rhizopogon spores can have an annoying tendency to be exactly 3µ wide when a couplet asks if they are greater or less than 3µ. In these cases, measure a number of mature spores (look for the fattest and skinniest ones); they will usually tend to range from either 2.8-3.0µ or 3.0-3.2µ. If in doubt, try both choices of the couplet and see which description fits best.
Genus Rhizopogon
1. Gleba marbled with white sterile tissue..... Genus Alpova
1. Not as above..... 2
2. Peridium with woven brown-walled hyphae in KOH (Fig. 2; an occasional piece of brown hyphae in
an otherwise hyaline peridium does not count); associated with Douglas-fir..... Section Villosuli
2. Not as above..... 3
3. Spores amyloid (parts of spores or spore walls darkening purplish or bluish-greenish in Melzer's iodine reagent;
Fig. 7)..... Section Amylopogon
3. Not as above..... Section Rhizopogon
Section Villosuli
All Pacific Northwestern species in this section associate only with Douglas-fir. The peridium is composed of loosely or densely woven
brown-walled hyphae (as viewed in KOH mount; Fig. 2). These hyphae are not apparent everywhere in
the peridium, but rather are in patches or clusters. This characteristic is best observed in peridial sections (plural
emphasized!) taken from different places around the peridium.
Often the mount needs to be squashed to separate out the hyphae from the dark compact peridial weave. Several species
(R. hawkerae, R. parksii, R. villescens, R. villosulus, and R. zelleri) have varying amounts of flagellate
hyphae that extend outward from the peridium. These hyphae are about 25-100µ long, 1-3µ wide at base, and end in a tapered tip
(Fig. 9).
1. Most spores ≥3µ wide..... 2
1. Most spores ≤3µ wide..... 5
2. Spores 5.5-8 x 3-4.5µ, more ellipsoid than cylindrical, truncate, individually pale green in KOH, brown
in mass, tramal hyphae having an ice-like appearance, brown-walled peridial hyphae patchy, subcutis often pink in KOH,
flagellate hyphae scant or absent, gleba yellow when young..... R. vinicolor (67) (=R. vesiculosus)3
2. Not as above..... 3
3. Spores club-shaped (thicker on one end), 8-13 x 4.5-7µ, peridial hyphae not always as
darkened as other Villosuli..... R. subclavitisporus (53) (also see R. hysterangioides)
3. Not as above..... 4
4. Spores 9-12 x 3.5-4µ, peridial sections without red tints, occasional flagellate hyphae, subhymenium filamentous..... R. zelleri (75)
4. Spores 7-10 x 3-4µ, peridial sections with red tints, copious flagellate hyphae, subhymenium cellular..... R. villescens (74)
5. Peridium very dark, spores 6-7 x 2.5-3, gleba charcoal-grey when dry and entirely green in KOH (not just greenish
patches)..... R. ater (sp nov.)
5. Not as above..... 6
6. Spores 4.5-6.5 x 2.3-3µ (short & fat)..... R. parksii (76)
6. Not as above..... 7
7. Subcutis with a red to pink layer in KOH (use 25x objective; Fig. 8)..... 8
7. Subcutis without a red to pink layer in KOH (may be orangish-brown), peridial hyphae large and loosely woven,
spores 6-8 x 2-2.5µ..... R. villosulus (84)
(=R. colossus)3
8. Spores 6.5-8 x 2.2-2.8µ, with occasional to abundant flagellate hyphae.....
R. hawkerae (83)
8. Spores 6-7 x 2-2.3µ, without flagellate hyphae ..... R. subareolatus (81)
Notes about R. hawkerae, R. subareolatus, and R. villosulus: The reddish subcutis characteristic of R. hawkerae and R. subareolatus fades with age into a pale pink band (after about 5 years of dried storage) and may disappear entirely after 7-10 years. R. subareolatus and R. villosulus can have occasional hyphal segments inflated to 20µ in the peridium. All of these species may have greenish tints to the gleba in KOH (particularly near the subcutis), but none to the extent of R. ater. It is possible that two or all of R. hawkerae, R. subareolatus, and R. villosulus are a single variable species. A phylogenetic analysis of this section is needed to clarify their relationships.
Section Amylopogon
The species in this section are typified by spores that when mounted in Melzer's solution appear partially bluish or
purple-grey (Fig. 7) or their walls darkened or "bolded". A quick way to check is to drop shavings
of gleba into a drop of Melzer's on a slide, if they are Amylopogon they will instantly turn black. All Pacific
Northwestern species of Amylopogon have peridia of rhizomorphic cables (Fig. 1), and the spores tend
to hang onto the basidia giving them a clustered appearance. Species with weak amyloid reactions are also keyed under other
sections.
1. Spores at least partially violet or spore walls appearing thick and dark in Melzer's..... 2
1. Spore walls pale blue or greenish in Melzer's..... 4
2. Spores 7-11 x 2-3.5µm, regularly shaped, spore walls darkening in Melzer's progressively as
they mature, many 100% darkened..... R. subpurpurascens (40)
2. Not as above.... 3
3. Spores 6-8 x 3-4.5, angular/irregularly shaped, walls of mature spores darkening in Melzer's
.... R. atroviolaceus (37)
3. Spores 6.5-10 x 3-5µ (usually short and fat), 25-75% (seldom 100%) of spore interiors partially to fully darkening in
Melzer's (Fig. 7), peridium drying dark brown to black, gleba drying bone-hard and difficult to
section..... R. milleri (35) (=R. kauffmannii)1
4. Spores 6-9 x 2-3.5µ, peridium thick (~300µ) and felty, of rhizomorphic cables (Fig. 1).....
R. salebrosus (37)
4. Spores 6-9 x 2.5-4µ, peridium thin (~100µ) and shiny, of woven hyphae (Fig. 3) but often with
embedded debris (Fig. 4)..... R. ellenae (43) (=R. subcaerulescens)1
Section Rhizopogon
Section Rhizopogon is where all species that do not fit the other sections are relegated. Whether or not the peridium of
a specimen stains or bruises red when fresh is an important characteristic, unfortunately this can be difficult to divine if
not explicitly stated in the fresh notes. Sometimes reddish tints can be discerned in the peridium of dry specimens, but
their absence cannot be construed as evidence that the fresh staining reaction did not occur.
1. Peridium reddish or bruising red when handled..... 18
1. Peridium not reddish or bruising red, or bruising reaction unknown..... 2
2. Peridium bright yellow, usually found in yellow mat, spores 7-10 x 3.5-5µ..... R. truncatus (57)
(=R. cokeri)3 (if columella present it is probably Truncocolumella citrina)
2. Not as above..... 3
3. Peridium very dark gray, spores 6-7 x 2.5-3, gleba charcoal-grey when dry and entirely green in KOH (not just greenish
patches)..... R. ater (sp nov.)
3. Not as above..... 4
4. All spores ≥3.5µ wide..... 5
4. At least some spores ≤3.5µ wide..... 8
5. Spores 11-14 x 4.5-6.5µ, football-shaped and often with an oil droplet, peridium of woven hyphae sometimes turning pinkish
in KOH mount, sporocarp small and knobby..... R. hysterangioides (50) (also see R. subclavitisporus)
5. Not as above..... 6
6. Spores 9-13 x 3.5-5µ, peridium of hyaline woven hyphae (Fig. 3), usually montane..... R. abietis (93)
6. Not as above..... 7
7. Spores 7-8 x 5-5.5µ, sterigmata very long (often >5µ), gleba of mature specimens drying dark chocolate
brown ..... R. exiguus (57)
7. Spores 8-10 x 4.5-6.5µ, relatively rare..... R. lutescens (62)
8. At least some spores >2.5µ wide (often ≥3µ; measure fattest ones)..... 9
8. All spores ≤2.5µ wide..... 12
9. Peridium pale yellowish with prominent rhizomorphic cables (macroscopically visible on peridium surface),
spores 5.5-7 x 2-3µ, with pines, frequently coastal, sporocarps often >25mm diameter
with orangish tints..... R. occidentalis (137) (also see R. ochraceorubens)
9. Not as above..... 10
10. Spores 6-9 x 2-3.5µ, peridium thick (~300µ) and felty, of rhizomorphic cables (Fig. 1).....
R. salebrosus (37)
10. Not as above..... 11
11. Spores 6.5-8 x 2.5-3µ, peridium and gleba drying dull cinnamon-brown, with pines..... R. separabilis (119)
11. Not as above (may have bruised red when fresh)..... 18
12. Peridium shiny black when dry..... 13
12. Not as above..... 14
13. Spores 5-7 x 1.8-2.5µ, peridium with brown pigmented balls in Melzer's..... R. brunneiniger (146)
13. Spores 5-7 x 2µ, peridium without brown pigmented balls in Melzer's..... R. nitens (147)
13. Not as above (may have bruised red when fresh)..... 21
14. Peridium ash-grey when dry, rosaceous in KOH, spores 5-6 x 2-2.3µ..... R. cinerascens (148)
14. Not as above..... 15
15. Peridium of woven hyphae (but may have some cable-like appressed hyphae), clean and free of pigmented debris, whitish with
apricot-salmon colored areas and usually drying pale, montane with Abies, spores 6-8 x 2-2.5µ..... R. subsalmonius (142)
15. Not as above..... 16
16. Peridium reddish-brown (yellow when young) with dark rhizomorphic cables wrapping surface (visible with hand lens; Fig. 10),
subcutis turning reddish in KOH (color change may take a minute), with pines, spores 6-8 x 1.7-2.5µ..... R. ochraceorubens (124) (=R. arenicola)3
16. Not as above..... 17
17. Peridium pallid when dry, spores 5-6 x 1.7-2µ..... R. bacillisporus (148)
17. Not as above (may have stained red when fresh)..... 21
18. At least some spores >3µ wide.....19
18. All spores ≤3µ wide.....21
19. Spores 6-9 x 2.5-4µ, peridium thin (~100µ) and shiny, of woven hyphae (Fig. 3) but often with embedded debris
(Fig. 4)..... R. ellenae (43) (=R. subcaerulescens)1
19. Not as above..... 20
20. Spores 5.5-8 x 3-4.5µ, more ellipsoid than cylindrical, individually pale green in KOH, brown in mass, tramal hyphae
having ice- or glass-like appearance, peridium of woven brown hyphae (Fig. 2), associated with
Douglas-fir..... R. vinicolor (67)
20. Spores 6.5-10 x 2.5-4.5µ, peridium of clear woven hyphae (Fig. 3), with pines..... R. roseolus (90) (=R. rubescens)2
21. Not as above..... 22
22. Peridium reddish-brown (yellow when young) with dark rhizomorphic cables wrapping surface (visible with hand lens; Fig. 10),
subcutis turning reddish in KOH (color change may take a minute), with pines, spores 6-8 x 1.7-2.5µ.....
R. ochraceorubens (124) (=R. arenicola)3
22. Not as above..... 23
23. Peridium of woven hyphae but often obscured by abundant reddish to orangish-brown pigmented debris ("chipboard" appearance;
Fig. 4), with only a few dark basal rhizomorphs, spores 5.5-8 x 2-3µ, with pines..... R. vulgaris (136)
23. Not as above, peridium structure readily apparent (without much pigmented debris), may have orangish or reddish tints..... 24
24. Peridium of woven hyphae (Fig. 3), usually drying dark, with scant pigmented debris, spores 6-8 x 2-2.3µ.....
R. evadens (151) (=R. smithii)3
24. Peridium of rhizomorphic cables (Fig. 1), usually drying dark, with some pigmented debris, spores
6.5-8 x 2-2.2µ..... R. evadens var. subalpinus (153)
24. Not as above..... 25
25. Use Smith & Zeller 1966 monograph (Memoirs of the New York Botanical Garden Vol. 14, No. 2) and subsequent updates, and good luck!
Footnotes:
1. Bidartondo MI & TD Bruns. 2002. Fine-level mycorrhizal specificity in the Monotropoideae (Ericaceae): specificity for
fungal species groups. Molecular Ecology 11:557-569.
2. Grubisha LC, JM Trappe, R Molina, & JW Spatafora. 2002. Biology of the ectomycorrhizal genus Rhizopogon. VI.
Re-examination of infrageneric relationships inferred from phylogenetic analyses of ITS sequences. Mycologia 94:607-619.
3. Personal communication, JM Trappe
Updates:
Smith AH. 1968. Further studies on Rhizopogon 1. The Elisha Mitchell Scientific Society 84(1):274-280.
Harrison KA & AH Smith. 1968. Some new species and distribution records of Rhizopogon in North America.
Can J Bot 46:881-899.
Miller SL. 1986. Hypogeous fungi from the southeastern United States 1. The genus Rhizopogon. Mycotaxon 72:193-218.
 |
 |
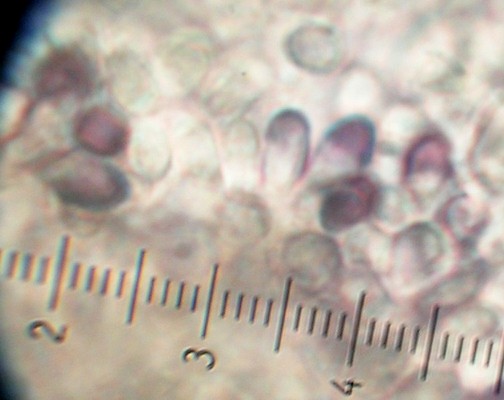
| Figure 7. Partially amyloid spores of R. milleri in Melzer's mount. 1000x. | Figure 8. Red peridial subcutis layer typical of R. hawkerae and R. subareolatus. 400x. |
 |
 |
| Figure 9. Flagellate peridial hyphae (arrow). 250x. | Figure 10. Appressed surface rhizomorphs of R. ochraceorubens. 10x. |